
According to the medication’s website, 9 million people in the US have been prescribed Chantix; a non-nicotine quit smoking medicine operating cheerfully under FDA approval. In comparison to e-cigarettes, the latter part of that sentence alone will be evidence enough to convince some people that the drug is the better of the two options. After all, FDA approval means it’s safe, right? And surely quitting smoking without relying on nicotine (as you do with e-cigs and nicotine replacement therapies) is better than continuing your addiction in some other form?
You might assume that Chantix must be effective, safe and well-studied to have FDA backing (particularly given the huge amount of fuss they've kicked up over e-cigarettes), but unfortunately the regulator’s approach to these quit smoking aids isn't quite as balanced as you’d hope. In fact, when you compare Chantix with e-cigarettes, pretty much the opposite picture emerges.
Contents
1 Chantix Vs. Electronic Cigarettes Comparison Chart
5 Randomized, Double-Blind, and Placebo-Controlled?
8 Conclusion: Chantix Vs. Electronic Cigarettes
Chantix Vs. Electronic Cigarettes
| Product | Chantix | E-Cigs |
|---|---|---|
| Purpose | Aid to smoking cessation treatment. How it works | Alternative to cigarettes |
| Cost | Around $360 for a three-month supply | $60 or less for a starter kit |
| FDA Approved | Yes | No |
| Form | Pill | Cigarette and Advanced Personalized Vaporizer |
| Success Rate | 44% | Learn more |
| Side Effects | Nausea; Insomnia; Abnormal dreams; Sleep disorder; Constipation; Gas; Vomiting; Suicidal thoughts. More on side effects | Throat irritation; Learn more. |
What is Chantix?
Chantix (or Champix outside the US) is the brand name for varenicline, a drug which basically works by preventing nicotine molecules from reaching the receptors in your brain by blocking the spaces. When you smoke, the nicotine stimulates these receptors and unleashes a dose of dopamine – the “reward” chemical in the brain responsible for the pleasurable feeling you get from sex, food, and many drugs of abuse – basically making you feel awesome for a short while. Chantix latches onto the nicotine receptors instead, meaning that smoking has virtually no effect. Although it is worth noting that Chantix releases some dopamine too, as a result of attaching to the happy-inducing nicotine receptor.
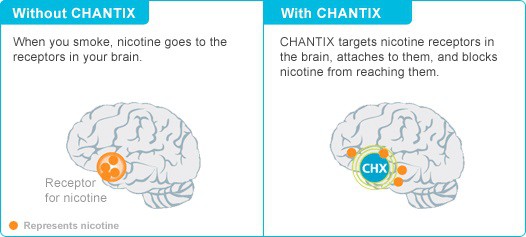
One of the most unique elements of Chantix is a stop-smoking medication is that users are actually encouraged to continue smoking for the first seven days of treatment. This gives the drug a chance to build up in their system, and eventually the nicotine will stop having an effect, in line with the nicotine-blocking action of the medicine. In other words, the cigarettes will cease to be pleasurable after a while on the medicine, anyway.
In comparison, electronic cigarettes obviously work in the more direct way. By using extracted pharmaceutical-grade nicotine, electronic cigarettes give you access to the nicotine without the huge quantities of harmful chemicals you find in cigarette smoke. This means that your receptors get stimulated just like they ordinarily would. Ultimately, the effect a chemical has is related to its structure, so even though varenicline can fit into the same “keyhole” as nicotine, the differences in structure mean that it wouldn't have identical properties.
Cost of Chantix
A three-month supply of Chantix (the recommended time-scale for those taking the drug) costs around $360, and it’s also important to factor in the expense of the initial seven days of smoking. This is equivalent to the amount you spend on cigarettes in a whole week. The drug is covered by some insurance providers, but the expense is still notable.
Comparatively, e-cigarette starter kits are available for $60 (or even less), leaving you with around $300 to spend on cartridges or liquids before you’ve even matched the cost of Chantix. Cartridges are more expensive, but you can still get 80 for $119.95, for example, or indeed just five for less than ten dollars. On that basis, you can get a starter kit plus around 190 cartridges for the same cost as a three-month supply of Chantix. Most vapers will get through a cartridge a day, but even if you use two cartridges per day, this works out to 95 days’ worth of vaping – longer than the three months offered by Chantix. It’s also worth mentioning that in a starter kit, you’ll usually get 10 cartridges, which works out to five days worth of heavy vaping.
How Effective is Chantix?
The marketing materials pushed by Pfizer, the manufacturer of Chantix, claims a 44 percent success rate after three months. This sounds very promising indeed, but unfortunately this figure is entirely misleading for several reasons. The most crucial point to understand is that three months is an inadequate amount of time on which to base a study of smoking cessation. This is particularly true because the treatment period of Chantix is three months, meaning that the study participants were taking the drug (which literally prevents nicotine from having an effect) for the entire period in which 44 percent of them managed to maintain abstinence from smoking. There is obviously a problem here, but unfortunately this is just the tip of an ugly, rotten iceberg.
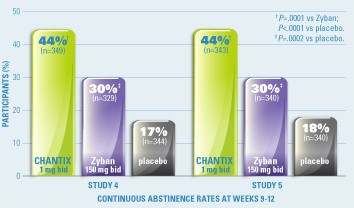
Chantix claims a 44% success rate after 3 months.
You might be wondering why the researchers of this particular study didn't take things any further, testing how effective Chantix was on overall smoking abstinence rates after a more meaningful interval (and, importantly, enough time for the drug to actually clear their system). They did, but you won’t find out about that if you look on Chantix’s webpage – in fact, you won’t even find information regarding which study they are actually talking about either. The reason is that over the course of a year, the abstinence rate dropped very significantly, down to just 23 percent. This is still twice as effective as the placebo (an inactive sugar solution), at 10.3 percent (and bupropion, an alternative medicine – at 14. 6 percent), granted, but there are many more problems with the research which means they shouldn't even be allowed this small victory.
Randomized, Double-Blind and Placebo-Controlled?
If you’re a doctor or even a researcher looking to find out whether Chantix is safe, these three phrases are what you’re looking for in a study’s methodology. In short, it means that the participants haven’t been screened to create an unduly negative or an unduly positive result for a particular treatment (as in, the participants are representative of the target population), that the results definitely weren't related to the placebo effect (if the group taking inactive sugar-pills do as well as those on the medicine, you know it’s something else creating the positive effect) and that neither the researchers nor the participants themselves knew who received the medicine and who received the placebo. Unfortunately, all of these three, confidence-building comments about the design of the study aren't completely warranted.
First of all, the criteria for inclusion in the study is so long that it isn't even worth replicating the entire list here to show how ridiculous it is. The paragraph explaining the grounds under which participants would be ineligible is about as long as the two paragraphs you've just read – over 250 words. Some of this is expected (after all, you don’t want a pre-existing ailment to make you think there’s a serious side effect, for example), but some of the groups excluded appear to have been driven quite notably by the difficulties ordinarily experienced in helping them quit smoking. Some of those who were excluded were under 18s, anyone suffering from depression, psychosis, bipolar or panic disorder, anybody smoking under 10 cigarettes per day, anybody who struggles with alcohol abuse and anybody who’s relapsed after trying nicotine replacement therapy or Zyban to try to quit recently.
The last one is obviously terrible, since a failed quitting attempt obviously represents a harder habit to break, and unfortunately many smokers relapse before being able to quit. However, there are problems with many of the others too. For example, teens are notably difficult to get to quit smoking, and almost half of smoking relapses can be attributed to alcohol consumption. This is obviously problematic, and according to WhyQuit.com, the majority of the groups excluded actually represent populations which are more challenging to help to quit. It was technically randomized according to the eligible participants, but those participants were unrepresentative of the smoking population. This means that the results for them don’t necessarily work for everybody.
On that note, can you afford private, one-to-one counseling with telephone support when you’re trying to quit smoking? Nope, neither can most people. But that didn't stop Pfizer from providing this to the participants of the study. This is another sure-fire method of creating an unduly positive result which doesn't reflect the majority of the target population.
The other two claims are ruined when you learn that around 75 percent of those receiving Chantix knew they were on the real medicine and 61 percent of those on the placebo knew that they were on the placebo. That means that the study (and the others Pfizer conducted) was actually not double blind, since the participants generally knew which group they were in, and not placebo-controlled, since placebos work on the assumption that they’re actually medicine.
In other words, the placebo group knew that they weren't taking the real thing, so they most probably wouldn't experience the placebo effect (where the “idea” of medicine is enough to produce a positive effect) and the group on the medicine may actually experience the placebo effect. The reason placebo groups are included is to ensure that the drug doesn't only work through the placebo effect, but in this study both elements were ruined, probably increasing the placebo effect amongst the Chantix group and decreasing it amongst the placebo group.
The Reality
The truth is that Chantix is actually about as effective as a nicotine patch, which is not very effective, both showing around a 33 percent success rate after a year in one particular study (which was also constructed with the same errors as those previously discussed). Generally, research on over-the-counter use is needed to reveal the true effectiveness of the medicine. For patches, this came in 2003, when a study revealed that only 7 percent of those who tried to quit using a patch failed to stay abstinent: “If the six patch trials are combined with the random effects model, the mean six month quit rate is also 7%.” This has yet to emerge for Chantix, but it appears to be following the same pattern as these initial treatments. It’ll seem effective when it’s still in the hands of the pharmaceutical companies, but when objective research comes out; we see that the real success rate is considerably lower.
For e-cigarettes, they've been studied by third party researchers numerous times, even with the odds stacked against the technology. There are many results which indicate the huge potential of e-cigs for helping people quit, but the most notable pitted the technology against smokers who didn't actually want to quit. In this study, the smokers tested had consumed over 15 cigarettes a day for over 10 years, and had no interest in stopping, but 55 percent of them were still able to either reduce their consumption by 50 percent or stop entirely after six months. On the whole, the group consumed 88 percent fewer cigarettes after using e-cigs over the course of half a year. Other studies have revealed statistics such as 67 percent of participants reducing their cigarette consumption after trying e-cigs and that they effectively reduce cravings and withdrawal symptoms.
The final argument to address regarding the effectiveness of Chantix vs. e-cigs is the idea that e-cigs “allow the addiction to continue” by still allowing the user to consume nicotine. This is why Chantix is so sanctimonious about being a “non-nicotine” medicine. It is true – of course – that you can stop smoking without relying on nicotine. However, you’d have to be living in a fantastical wonderland of your own creation if you thought that this worked for everybody. If it did, we wouldn't have nearly as many smokers today. E-cigs have potential to help people stop smoking, but they’re really about harm reduction.
In simple terms, this is reducing the risks to the individual even if the addiction technically continues. It’s a treatment philosophy that recognizes that abstinence isn't for everybody, since it obviously isn't. Despite that fact about electronic cigarettes, the 6-month study on smokers who didn't want to quit found that 22.5 percent were completely abstinent at the end of the study. You might argue that this doesn't count since they were using the e-cig and thus consuming nicotine, but one of the many tricks built into the Chantix studies is that those classed as “successfully” abstinent were actually freely allowed to consume nicotine replacement products – a term which technically describes e-cigs, in addition to gums and patches. In other words, even Chantix’s definition of abstinent still allows for nicotine consumption. This isn't a problem of course (comparatively safe nicotine consumption is much better than dangerous consumption), but it’s another way in which Chantix’s research isn't quite what it seems.
The Nail in the Coffin: Side Effects
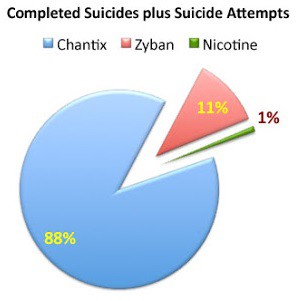
You can see that the benefits of Chantix are greatly exaggerated, but the side effects really drive the nail into their coffin. Whereas e-cigarettes have shown virtually no side effects aside from throat irritation (and actually reduce the side-effects of smoking), Chantix appears to cause suicidal thoughts and acts in some users. You might recall that those with many psychological conditions were exempt from the clinical trials, which obviously either allowed the reaction to escape notice or was constructed to purposefully prevent it. The FDA added safety warnings and basically allowed the drug to continue being sold. Even the official website has to be plastered with warnings.
The other side effects from Chantix include:
- Nausea (around 30 percent experience this)
- Insomnia (18 percent)
- Abnormal dreams (13 percent)
- Constipation (8 percent)
- Sleep disorders (5 percent).
These are quite mild effects, and it is worth noting that the serious psychological adverse reactions are fairly rare, although you’re advised not to take it if you have an existing condition. For a personal account of Chantix-madness, read this.
It doesn't get better from there either: a 2011 study revealed that those taking Chantix are at increased risk of cardiovascular events such as heart attacks and irregular heartbeat. The numbers of adverse events reported by the study are low, but when the statistics were churned the researchers concluded that “The use of varenicline [Chantix] among tobacco users was associated with a 72% increased risk of serious adverse cardiovascular events.” This is obviously significant and extremely, extremely worrying.
Conclusion: Chantix Vs. Electronic Cigarettes
With a mounting list of serious side effects from Chantix and only more information regarding the effectiveness and lack of side effects of e-cigarettes surfacing, the decision is actually a pretty simple one. Do you want to take a potentially dangerous drug, peddled in the most misleading fashion possible by a monolithic corporation? Chantix is a revolutionary idea, but reality is now creeping in and showing that things aren't as pleasant as they seem. In fact, if you consider the cost, the quality of the supporting research, the respective risks, the potential for harmful effects and the effectiveness of both, there really is only one choice. E-cigs wipe the floor with Chantix.

